The atom is the fundamental building block of everything we see, touch, and breathe. From the screen you are reading this on to the air in your lungs, everything is made of these incredibly tiny units of matter.
Table of Contents
What is an Atom?
An atom is the smallest unit of an element that retains the properties of that element. While the word “atom” comes from the Greek word atomos, meaning “indivisible,” we now know that atoms are made of even smaller subatomic particles.
Subatomic Particles
Every atom (with the exception of some hydrogen) is composed of three primary particles:
Protons:
- Charge: Positive (+1)
- Location: Inside the nucleus (the center).
- Role: The number of protons defines the identity of the element (e.g., every Carbon atom has 6 protons).
Neutrons:
- Charge: Neutral (0)
- Location: Inside the nucleus.
- Role: They act as “glue” to stabilize the nucleus, preventing the positive protons from repelling each other.
Electrons:
- Charge: Negative (-1)
- Location: Orbiting the nucleus in “shells” or “clouds.”
- Role: Electrons are responsible for chemical bonding and electricity.
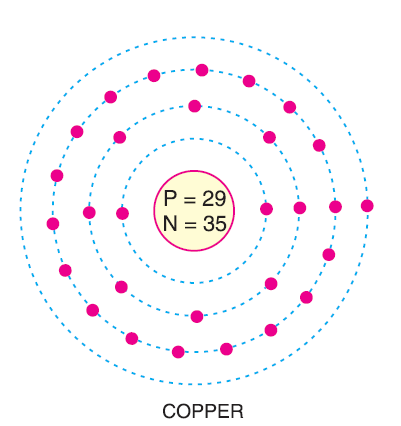
Atomic Structure
To visualize an atom, think of it in two main parts:
The Nucleus
The center of the atom is a dense, tiny core called the nucleus. Even though the nucleus contains 99.9% of the atom’s mass (protons and neutrons), it takes up only a tiny fraction of the atom’s actual volume.
The Electron Cloud
Electrons don’t orbit the nucleus like planets around the sun in perfect circles. Instead, they exist in a “cloud” or specific energy levels. The further an electron is from the nucleus, the more energy it has.
In the context of atomic structure, these two numbers are the “ID card” of any element. They tell you exactly what the element is and how much it weighs.
Atomic Number ($Z$)
The Atomic Number is the most important characteristic of an atom. It is the count of protons in the nucleus.
- Identity: It defines the element. For example, any atom with 6 protons is always Carbon. If you change the number of protons, you change the element itself.
- Electrical Balance: In a neutral (uncharged) atom, the Atomic Number also equals the number of electrons.
- Periodic Table: Elements are arranged in the periodic table in increasing order of their Atomic Number.
Atomic Mass Number ($A$)
The Atomic Mass Number (or Nucleon Number) is the total count of particles inside the nucleus.
- Formula: $A = \text{Protons} + \text{Neutrons}$
- Weight: Electrons are so light that their mass is ignored; therefore, the mass of the atom is almost entirely concentrated in the protons and neutrons.
- Variation (Isotopes): While the Atomic Number ($Z$) stays the same for an element, the Mass Number ($A$) can vary if the number of neutrons changes. These variations are called Isotopes.
Comparison at a Glance
| Feature | Atomic Number ($Z$) | Atomic Mass Number ($A$) |
|---|---|---|
| Represents | Number of Protons | Protons + Neutrons |
| Location | Bottom-left of symbol ($_6C$) | Top-left of symbol ($^{12}C$) |
| Purpose | Identifies the element | Determines the mass/isotope |
| Changeability | Never changes for an element | Can change (forming isotopes) |
Quick Calculation Tip
To find the number of neutrons in an atom, simply subtract the Atomic Number from the Mass Number:
$$\text{Neutrons} = A – Z$$
$$\text{Neutrons} = A – Z$$
Example: Sodium ($^{23}_{11}Na$)
- Atomic Number ($Z$): 11 (so, 11 protons)
- Mass Number ($A$): 23
- Neutrons: $23 – 11 = 12$ neutrons.