In an atom, the behavior of electrons is determined by their energy levels. To understand how materials conduct electricity, we look at the Valence Band and the Conduction Band.
Table of Contents
Valence Electrons
Valence electrons are the electrons located in the outermost shell of an atom. They are the furthest from the nucleus and are responsible for chemical bonding.
- Energy Level: They occupy the highest occupied energy band, called the Valence Band.
- Bonding: They are still “attached” to the atom. They jump between atoms to form molecules but stay within the structure of the material.
- State: At $0\text{ K}$ (absolute zero), the valence band is usually completely filled with these electrons.
Materials can be classified based on the number of valence electrons in their atoms. When the number of valence electrons is less than 4, the material is generally a metal and behaves as a conductor, such as sodium, magnesium, and aluminium.
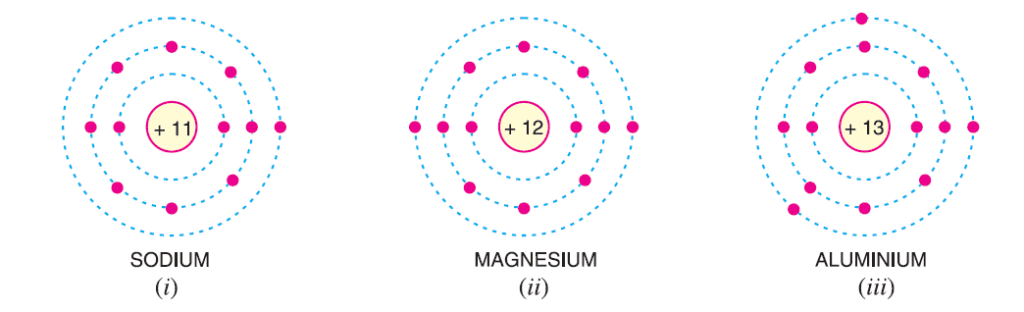
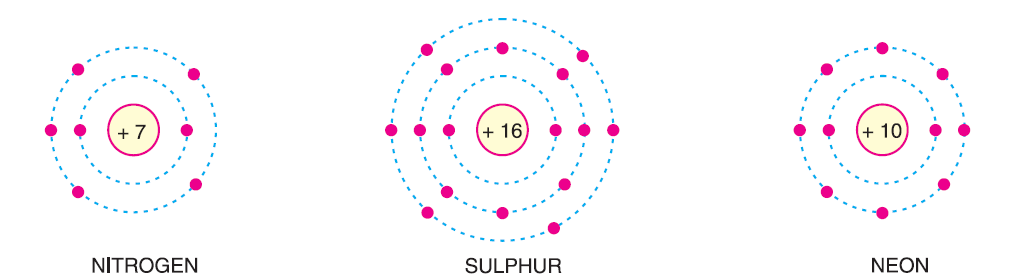
If the number of valence electrons is greater than 4, the material is usually a non-metal and acts as an insulator, as seen in Nitrogen, Sulphur, and Neon.
When the number of valence electrons is exactly 4, the material exhibits properties of both metals and non-metals and is typically a semiconductor, with common examples being carbon, silicon, and germanium.
Conduction Electrons
Conduction electrons are valence electrons that have gained enough energy (from heat, light, or an electric field) to break free from the attraction of the nucleus.
- Energy Level: They move into a higher energy level called the Conduction Band.
- Mobility: Once in this band, they are “free electrons.” They can move throughout the material and carry an electric current.
- Conductivity: The more electrons in this band, the better the material conducts electricity.
The Forbidden Energy Gap ($E_g$)
The “distance” between the Valence Band and the Conduction Band is called the Energy Gap. It determines whether a material is a conductor, semiconductor, or insulator:
| Material Type | Energy Gap Status | Behavior |
|---|---|---|
| Conductors (e.g., Copper) | Overlap | Electrons move easily from valence to conduction band. |
| Semiconductors (e.g., Silicon) | Small Gap | Some electrons jump the gap when heated or “doped.” |
| Insulators (e.g., Glass) | Large Gap | Electrons cannot jump the gap; no current flows. |